The 2025 Diversion Trends Report
The 2025 Diversion Trends Report offers a nationwide look at how hospitals are working to detect and prevent drug diversion. Drawing on 266 million controlled substance transactions from more than 1,100 hospitals, the report highlights current challenges, leading diversion indicators, and the impact of staffing and software investments. It provides a clear snapshot of today’s diversion landscape, showing trends in documentation accuracy, investigation workflows, and confirmed diversion cases across U.S. health systems.

Hospitals With Full-Time Diversion Monitoring Staff
75% of hospitals report having full-time staff dedicated to diversion monitoring, most commonly one or two individuals.
Transactions With Documentation Variances
6% of all controlled substance transactions contained documentation variances that required further review.
Hospitals With a Diversion Oversight Committee
67% of diversion-focused staff say their organization has a cross-functional diversion committee in place.
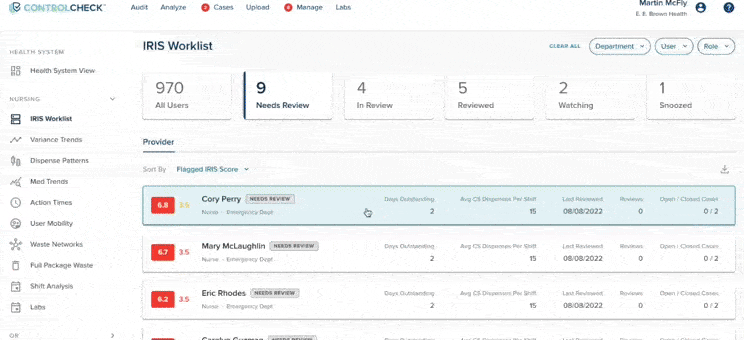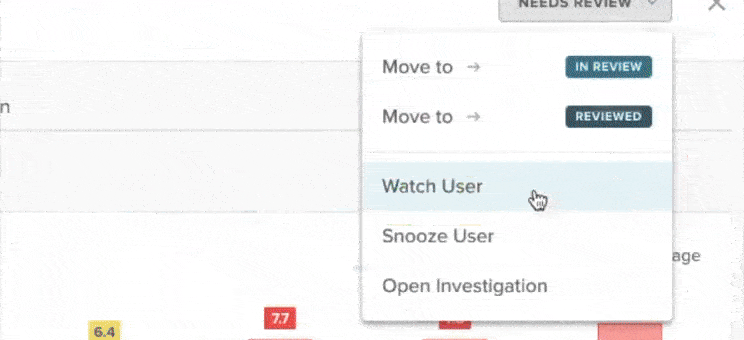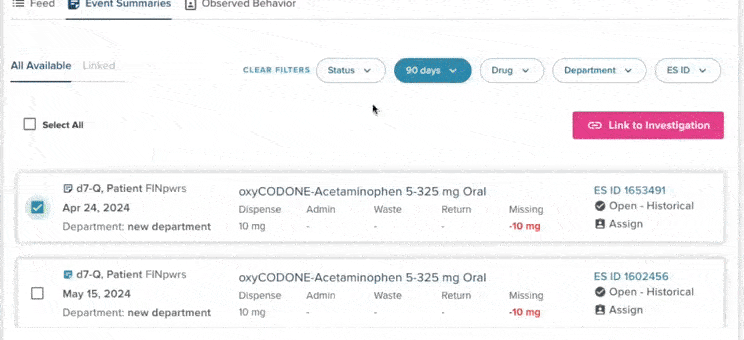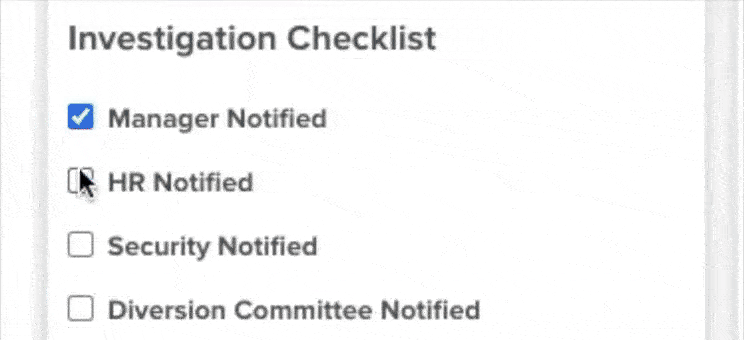
53% of diversion investigations are triggered by software alerts
More than half of all diversion investigations begin with automated data flags in ControlCheck. These alerts identify unusual behavior earlier than routine surveillance and help hospitals prioritize high-risk cases. Facilities increasingly rely on software to surface discrepancies that may otherwise go unnoticed.
48% of controlled substance variances stem from incorrect documentation
Incorrect documentation remains the most common cause of medication variances, including mischarting, wrong patient entries, and incorrect doses. These errors create operational burden for diversion teams and can obscure early signs of diversion. Improving documentation accuracy continues to be a top compliance priority.
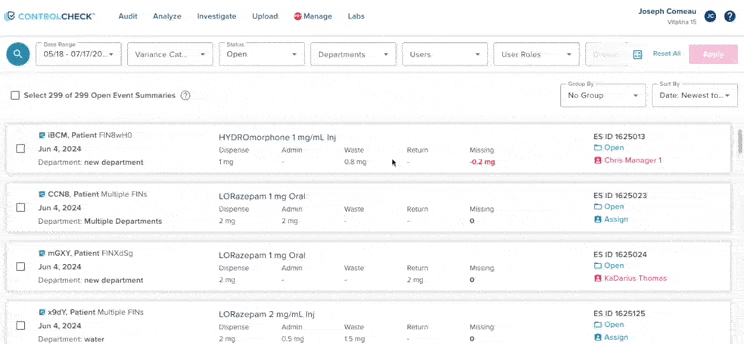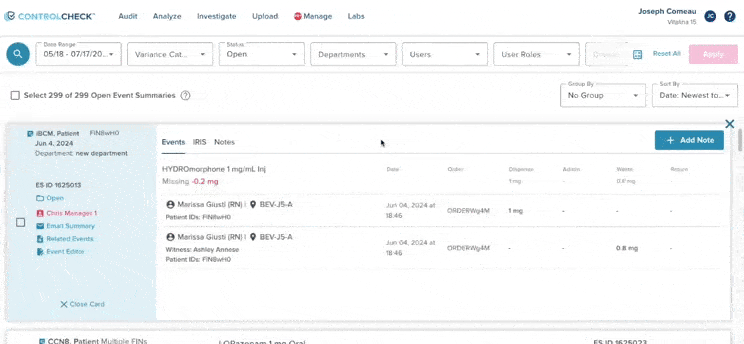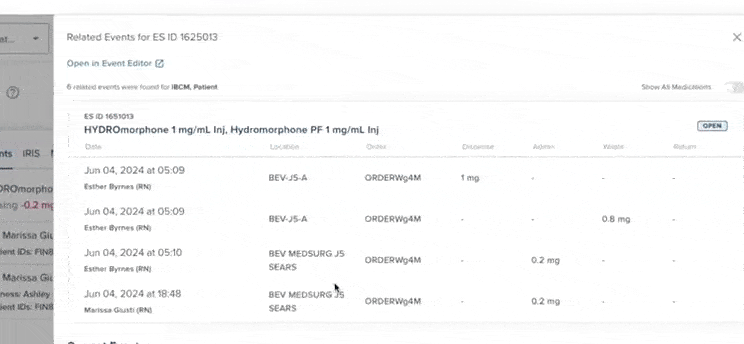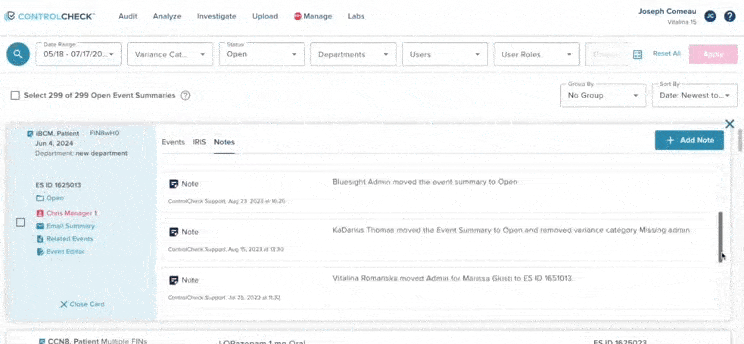
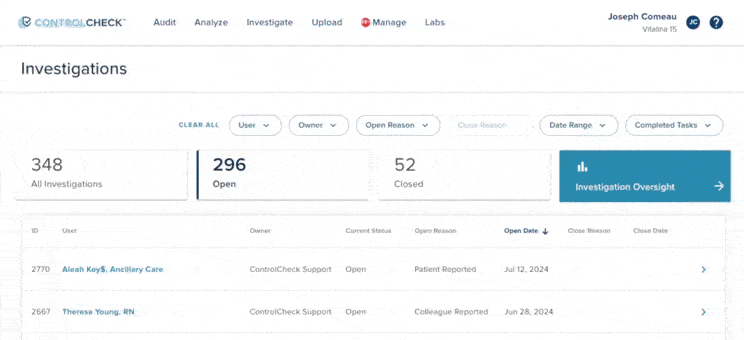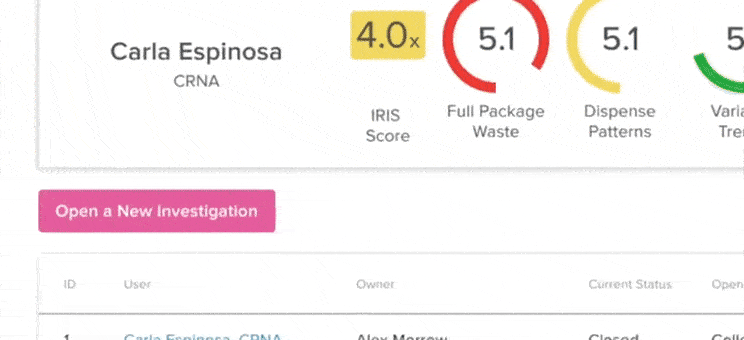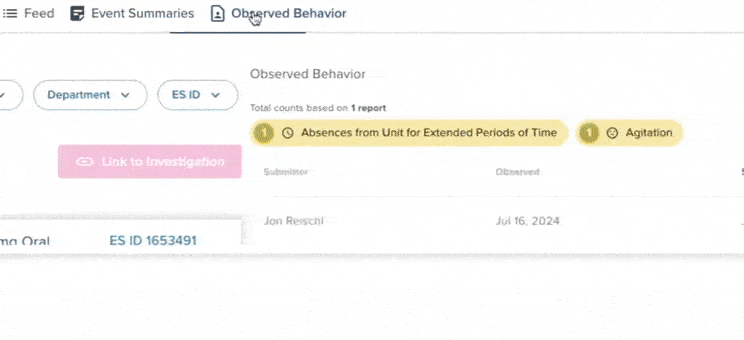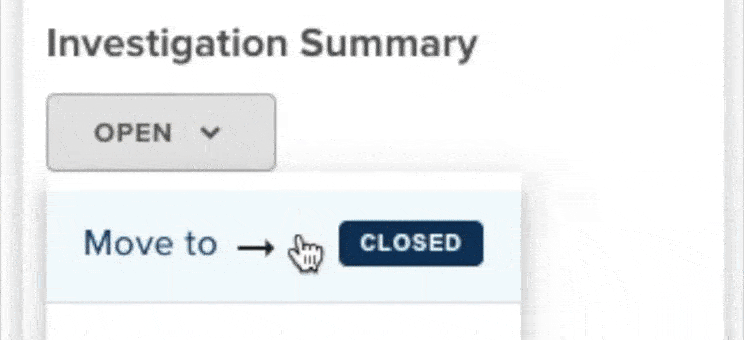
61% increase in diversion investigations since 2023
Hospitals are conducting more diversion investigations as monitoring workflows expand and variance detection improves. The average hospital now reviews significantly more cases each quarter. At the same time, investigations are closing faster due to more efficient review processes.



